Curriculum Links
The particulate nature of matter
– the properties of the different states of matter (solid, liquid and gas) in terms of the particle model, including gas pressure
– changes of state in terms of the particle model
Atoms, elements and compounds
– differences between atoms, elements and compounds
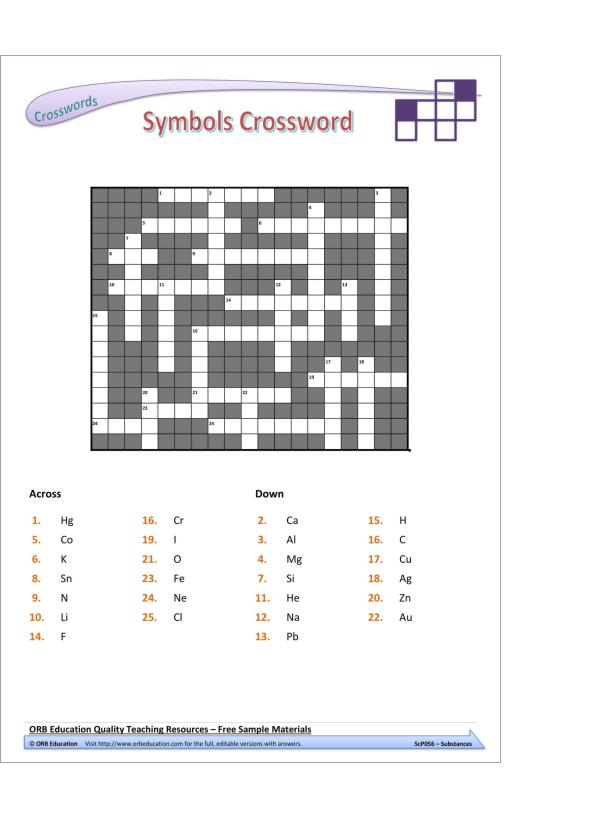
– chemical symbols and formulae for elements and compounds
– conservation of material and of mass, and reversibility, in melting, freezing, evaporation, sublimation, condensation
– similarities and differences, including density differences, between solids, liquids and gases
– Brownian motion in gases
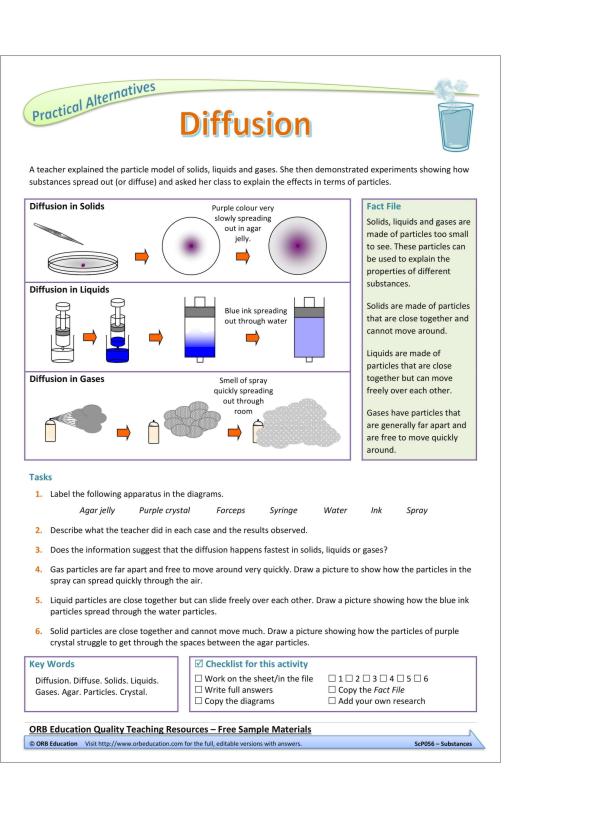
– diffusion in liquids and gases driven by differences in concentration
– diffusion in terms of the particle model
– the differences in arrangements, in motion and in closeness of particles explaining changes of state, shape and density;
– atoms and molecules as particles
– changes with temperature in motion and spacing of particles





Reviews
There are no reviews yet.